 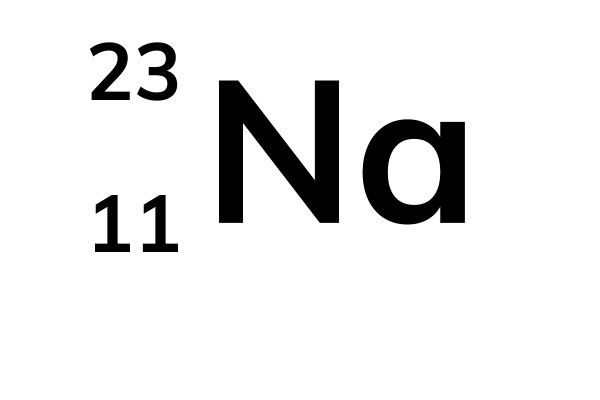
The amount of example an isotope makes up determines how much it contributes to the normal. The normal of the atomic masses of the seeming plurality of distinct isotopes in an example is the general atomic mass. Because the isotopes of an element have different atomic masses, researchers can determine the element’s overall atomic mass (also known as the atomic weight). 
In most cases, the atomic mass number is rounded to the next whole number. Carbon-12, for example, is a typical carbon atom with six neutrons and six protons. The absolute mass of a single atom is its atomic mass, which is measured in atomic mass units, or amu. Carbon-12 has 6 protons and 6 neutrons, carbon-13 has 6 protons and 7 neutrons, and carbon-14 has 6 protons and 8 neutrons. For example, carbon has three isotopes: carbon-12, carbon-13, and carbon-14. Isotopes are atoms of the same element that have different numbers of neutrons. It is typically expressed in atomic mass units (amu), which are defined as 1/12 of the mass of a carbon-12 atom.The atomic mass of an atom is a weighted average of the masses of the isotopes of that atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
:max_bytes(150000):strip_icc()/PeriodicTableSigFigBW-58b5c7f25f9b586046cae098.png)
 RSS Feed
RSS Feed
